|
In addition, during the binding process, the ion must be transferred from a high dielectric constant medium (water = ∼80) to a relatively low dielectric constant environment (protein = ∼4). In some cases the unbound protein residues are solvated prior to binding as well, which only contributes to the energetic barrier. 2 – 4 These solvation energies arise from the classic ion–dipole interaction between ions and water dipoles. For example, the solvation energies of sulfate and potassium are estimated to be approximately −300 and −80 kcal/mol, respectively. 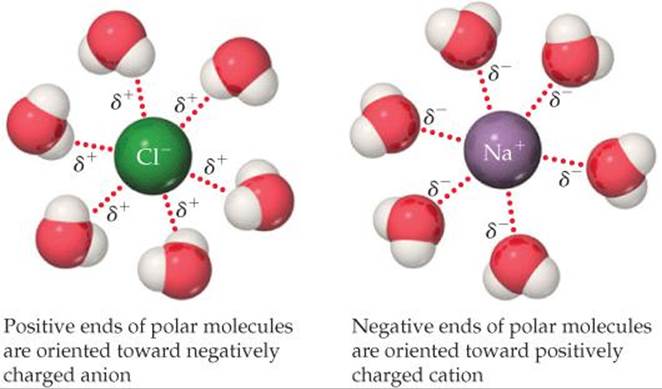
The ion–dipole interactions are sufficiently favorable to overcome the desolvation energies of these ions, which are frequently very high. Second, the charged atoms or ligands are completely desolvated and buried within the protein. (B) Helix N-termini in the sulfate-binding site, each providing a main chain NH dipole. Carbons are colored green nitrogen, blue oxygen, red sulfur, gold. Dashed lines represent ion–dipole interactions via hydrogen bonds, and cooperative hydrogen bonds are colored purple. Salmonella typhimurium SBP-sulfate complex X-ray structure (PDB ID 1sbp). In this brief review, we will define ion–dipole interactions and discuss several diverse functional roles they play in biological and biochemical processes. 1 In this instance, ion–dipole interactions govern charged ligand specificity, desolvation, and charge compensation. This changed significantly with the X-ray structure of the sulfate-binding protein (SBP), the primary high-specificity, high-affinity sulfate ABC transport receptor, which revealed that the sulfate was completely buried, desolvated, and bound solely by neutral dipolar groups (described subsequently). Originally, conventional wisdom stated that ions interacted solely through charge coupling with counterions or salt links with countercharged residues in proteins. In biological macromolecules, these ubiquitous interactions and their roles in protein function are largely underappreciated. They encompass a broad number of more specifically defined interactions, because they include charge–neutral hydrogen bonds and coordinations. Which is the main difference between intramolecular bonding and intermolecular bondings like dipole-dipole and hydrogen bondings.Ion (charge)–dipole interactions are fundamental attractive forces, akin to hydrogen bonds and Van der Waals interactions, that are formed between ions or charged molecules and dipolar groups with partial charges. Note: Both dipole-dipole and hydrogen bonding happen between atoms of two different molecules and not between atoms of the same molecules. 
With such a high difference in the electrostatic behaviour and such strong polarity, the bonding becomes extra strong and thus Hydrogen bonding is stronger than dipole-dipole interactions. When the partially positive y charged hydrogen atom comes in contact with a neighbouring partially negatively charged fluorine, nitrogen or oxygen atom, hydrogen bonding takes place.Īs hydrogen is a special case of Dipole-dipole interactions and we know that it is an electrostatic attraction, the hydrogen bonding becomes the strongest of all dipole-dipole interactions as the fluorine, nitrogen or oxygen atoms are much more electronegative than hydrogen which makes the polarity of the bond extra strong. Such type of bonding happens especially when a hydrogen atom is bonded to either a nitrogen, oxygen or fluorine atom. Hydrogen bonding, on the other hand, is a special type of dipole-dipole bonding. For such bondings to happen, the ions must be charged. Hydrogen bonding and dipole dipole interactions are both intermolecular in nature.ĭipole-dipole interactions generally occur when partially negatively charged molecules interact with a neighbouring molecule that is positively charged. Hint:Before determining the strength of each type of bonding, it is important to know that there are two types of attraction in between molecules and are intramolecular and intermolecular.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
